 
This is just the difference in entropy between the sum of the products and the sum of the reactants.Įxample: Calculate the entropy change for this reaction:ġ. The formula for calculating ΔS is as follows: 
They are typically found at standard conditions. 
In a chemical reactions, products and reactants have different entropies – there is an entropy change.įor reactions with a negative entropy change, reactants will have a higher entropy than products.įor reactions with a positive entropy change, products will have a higher entropy than reactants.Įntropy values have been determined and can be found in databases. Therefore, substance will naturally move in order to increase their entropy. The more disordered a system is, the higher its entropy and the more stable it is. When in a reaction the number of moles of product formed is greater than the number of moles of reactants, there is an increase in entropy. Number of particles – For an increased number of particles there is and increased number of ways in which these can be arranged and therefore an increased entropy. Dissolving – When a solid dissolves in a solvent, its entropy increases because of the free movement of the dissolved particles.ģ. Thermodynamic – Introduction to EntropyĢ. This is because the arrangement of particles is more random in gases than solids. State of Matter – Generally, solids have lower entropy than liquids, which have lower entropy than gases. The more disordered a system is, the higher (the more positive) the value of entropy.ġ. Energy has spontaneously become more dispersed and spread out in that ‘universe’ than when the glass of ice and water was introduced and became a 'system' within it.Thermodynamic - Introduction to Entropy (A-Level Chemistry) Introduction to Entropy Entropy Key TermsĮntropy can be defined as the randomness or dispersal of energy of a system. this thermodynamic system, has increased in entropy. This is always true in spontaneous events in a thermodynamic system and it shows the predictive importance of entropy: the final net entropy after such an event is always greater than was the initial entropy.Īs the temperature of the cool water rises to that of the room and the room further cools imperceptibly, the sum of the δQ/T over the continuous range, “at many increments”, in the initially cool to finally warm water can be found by calculus. It is important to realize that the entropy of the surrounding room decreases less than the entropy of the ice and water increases: the room temperature of 298 K is larger than 273 K and therefore the ratio, (entropy change), of δQ/298K for the surroundings is smaller than the ratio (entropy change), of δQ/273K for the ice and water system. The heat δQ for this process is the energy required to change water from the solid state to the liquid state, and is called the enthalpy of fusion, i.e. 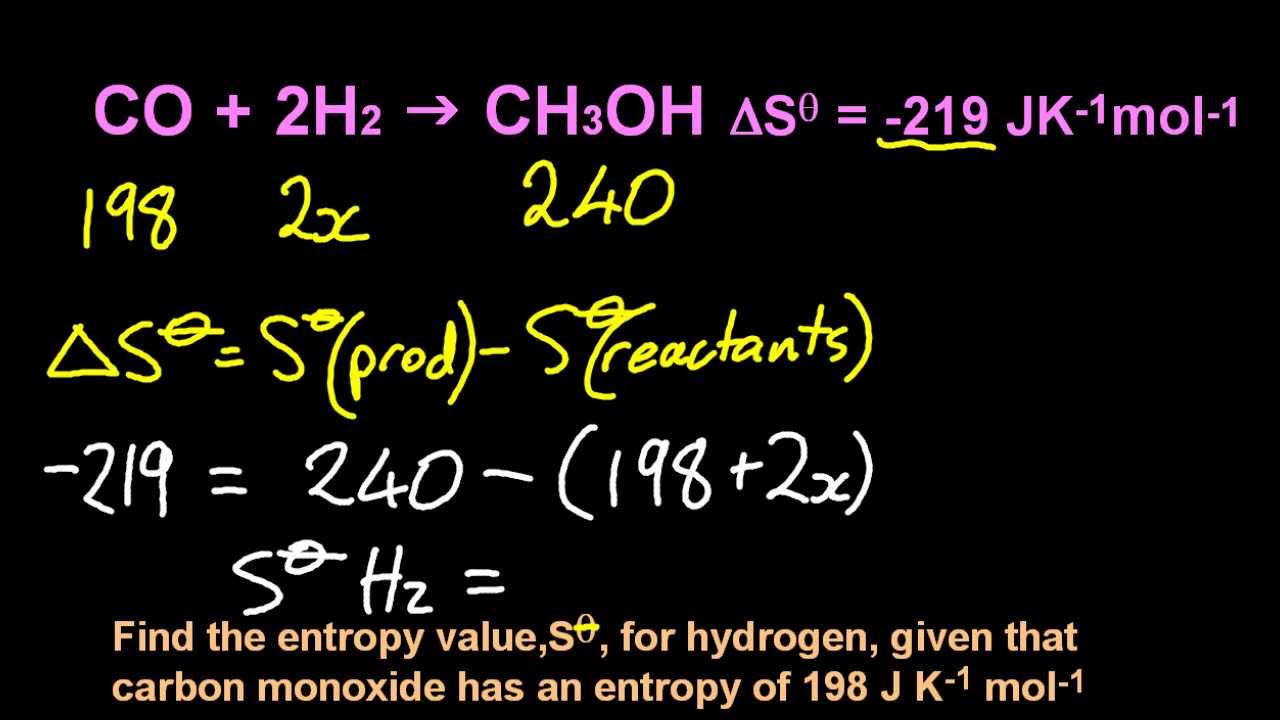
The entropy of the system, which is δQ/T, increases by δQ/273K. In this system, some heat ( δQ) from the warmer surroundings at 298 K (25 ☌ 77 ☏) transfers to the cooler system of ice and water at its constant temperature ( T) of 273 K (0 ☌ 32 ☏), the melting temperature of ice. Ice melting provides an example in which entropy increases in a small system, a thermodynamic system consisting of the surroundings (the warm room) and the entity of glass container, ice, water which has been allowed to reach thermodynamic equilibrium at the melting temperature of ice. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
